Therapeutic and diagnostic products
Femasys has achieved global regulatory approvals for a first-line infertility treatment and complimentary diagnostic products.
Infertility
Infertility has become a significant global problem with no affordable infertility options introduced in decades.

Key Trends
- 43 U.S. states have recorded lowest fertility rate in last three decades2
- 9.14 million women have received infertility services at some time in their lives3
- Centers for Disease Control and Prevention (2019)
- Chandra A, et al. (2014)


- Kumar N, et al. (2015) Trends of male factor infertility, an important cause of infertility: A review of literature. J Hum Reprod Sci. 8(4): 191–196. doi: 10.4103/0974-1208.170370:
- Levine H, et al. (2023) Temporal trends in sperm count: a systematic review and meta-regression analysis of samples collected globally in the 20th and 21st centuries. Human Reproduction Update, Vol.29, No.2, pp. 157–176, 2023
Our Infertility Portfolio
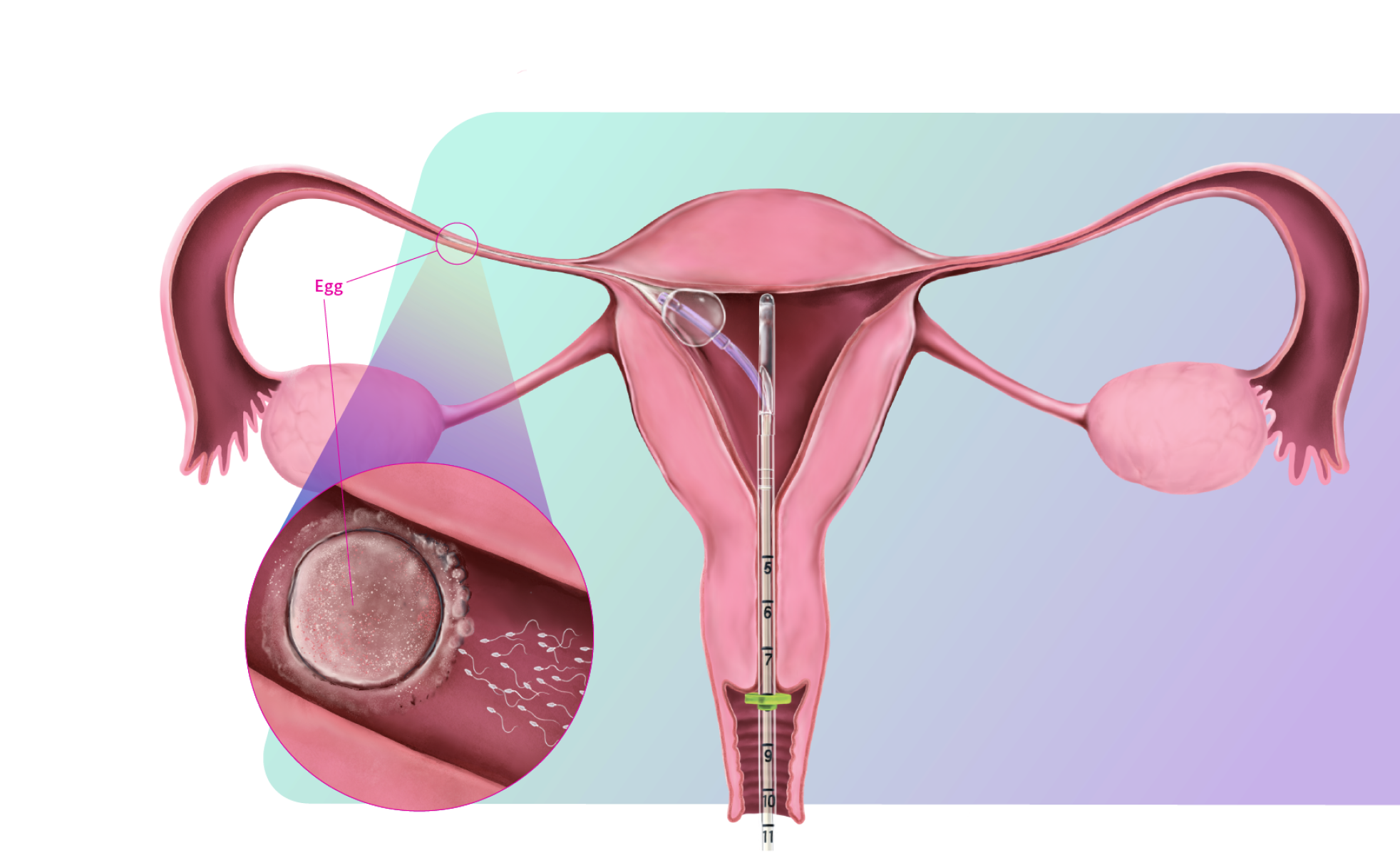
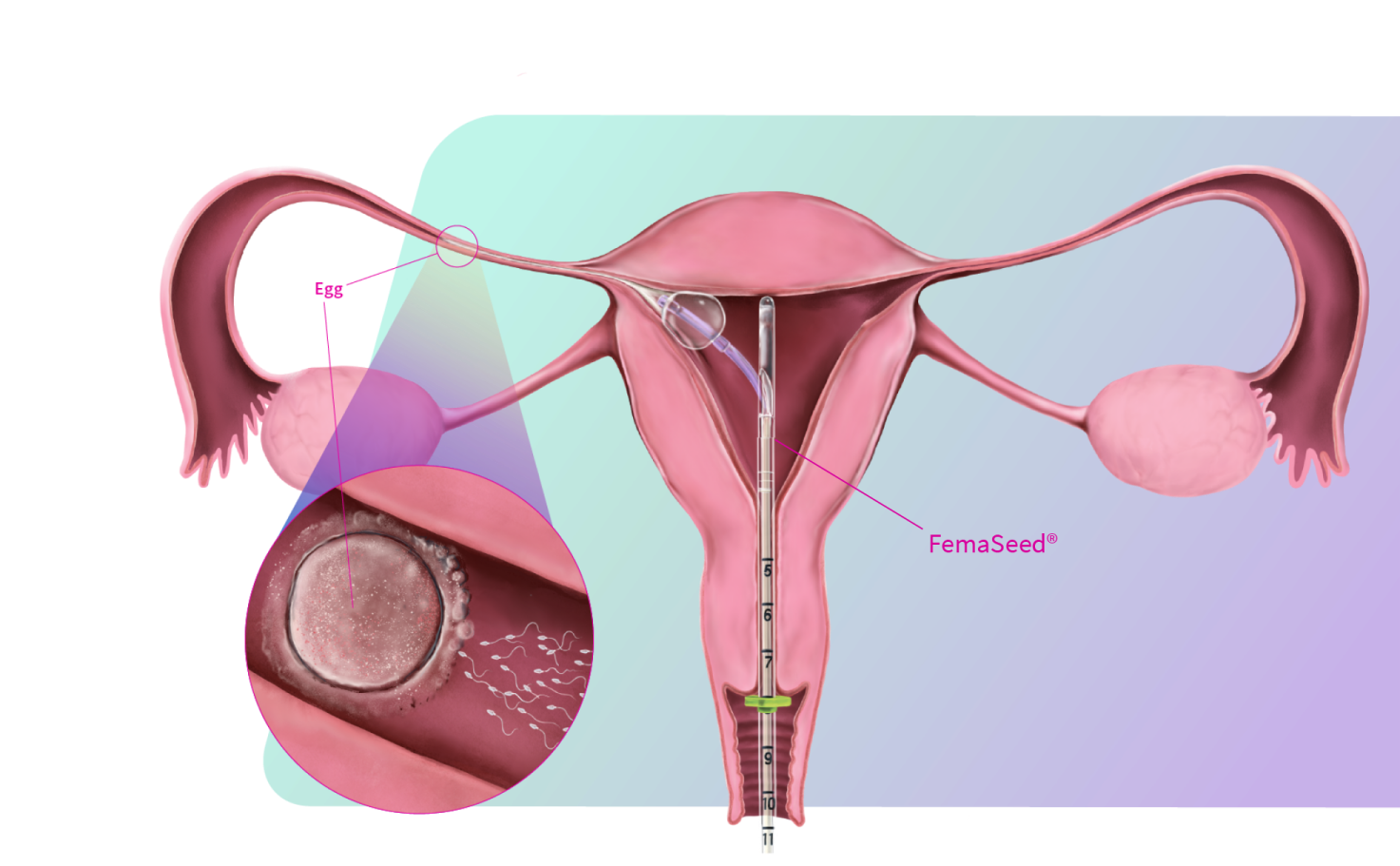
FemaSeed is the latest in artificial insemination technology, engineered to precisely deliver sperm into the fallopian tube, the site of conception, to enhance natural fertilization. As a highly cost-effective approach that carries notably reduced risks compared to in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI), FemaSeed is positioned as a first-line therapeutic option.
FemaSeed has regulatory approvals in the U.S., Europe, U.K., Canada, and Israel.
Clinical Data
Trial Design:
Prospective, multi-center, unblinded pivotal clinical trial with historical control. Conducted under FDA Investigational Device Exemption (IDE) approval. NCT0468847
Trial Results6:
Primary Endpoint
(for male-factor/unexplained fertility (1-20 million total motile sperm count, TMSC))
- Pregnancy rate was 26.3% by subject (n=38) and 17.5% by cycle (n=57)
Safety
- Adverse events were consistent with IUI.
- Liu, J. H., Glassner, M., Gracia, C. R., Johnstone, E. B., Schnell, V. L., Thomas, M. A., Morrison, L., Lee-Sepsick, K. (2024). FemaSeed Directional Intratubal Artificial Insemination for Couples with Male-Factor or Unexplained Infertility Associated with Low Male Sperm Count. J Gynecol Reprod Med, 8(2), 01-12.
- Duran HE, Morshedi M, Kruger T, Oehninger S. Intrauterine insemination: a systematic review on determinants of success. Hum Reprod Update. 2002;8(4):373‒84. doi: 10.1093/humupd/8.4.373
Market Research
Number of participants: 1,000 infertile women
- More than 7 in 10 women were extremely or very likely to consider insemination with FemaSeed
- For those who received IVF, 88% would have preferred FemaSeed prior to moving on to IVF
- 85% who received IUI would have preferred FemaSeed instead of IUI

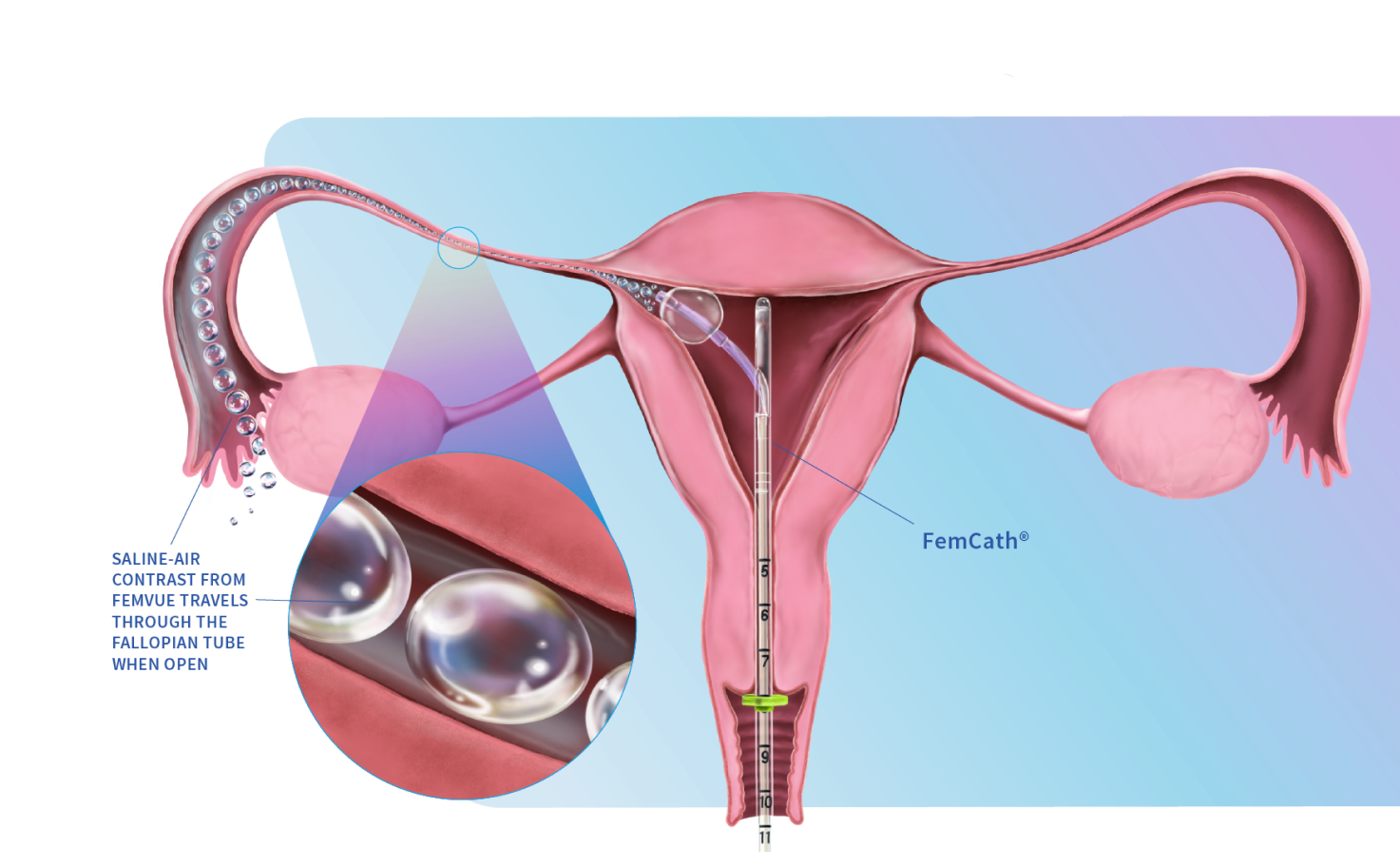
FemVue is the first FDA-cleared product that creates natural saline and air contrast and enables safe, reliable, and real time evaluation of the fallopian tubes with ultrasound. When performed with a uterine cavity assessment, a more comprehensive exam can be achieved from the comfort of the GYN’s office.
FemVue has regulatory approvals in the U.S., Europe, U.K., Japan, Canada, and Israel.

FemCath, an FDA-cleared intrauterine catheter, is the first to allow for selective evaluation of a fallopian tube with contrast. FemCath features our proprietary delivery platform, which places balloon technology close to the opening of a selected fallopian tube for directed delivery.
FemCath has regulatory approvals in the U.S., Europe and Canada.